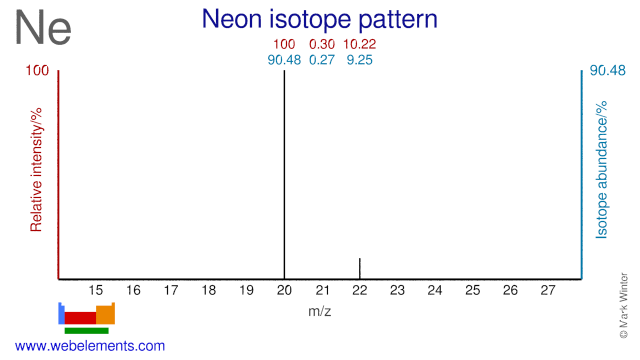
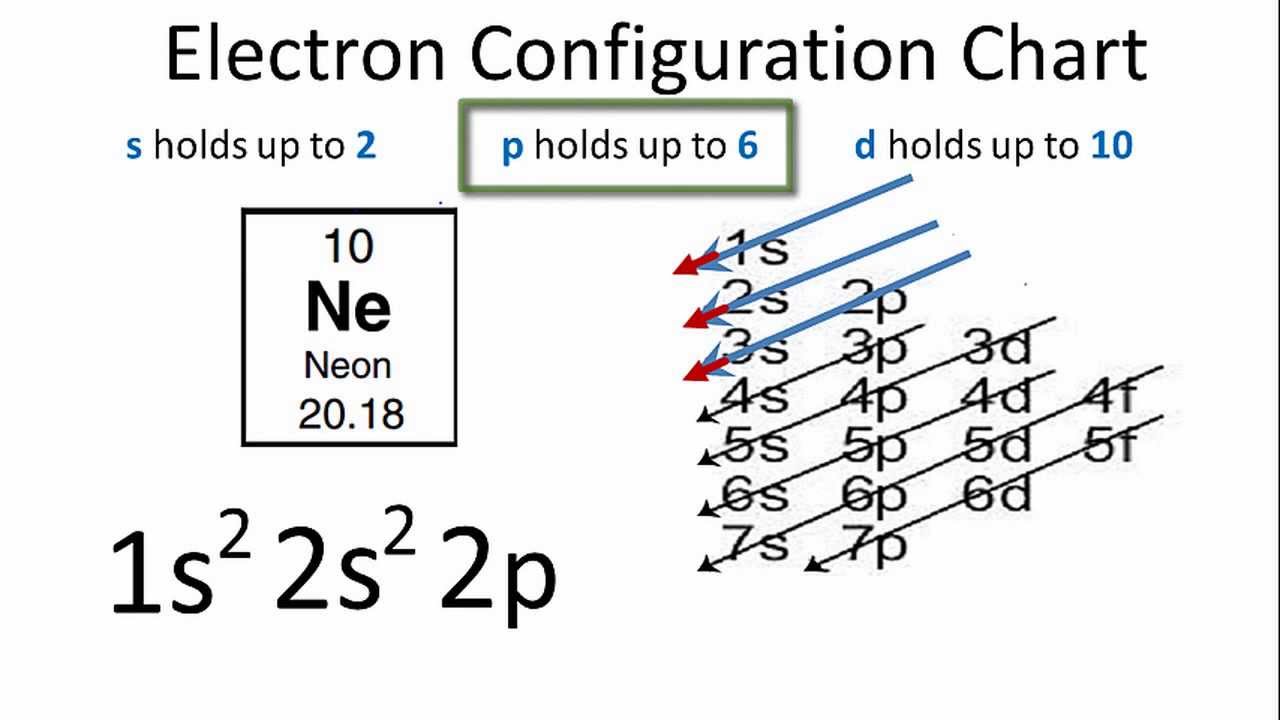
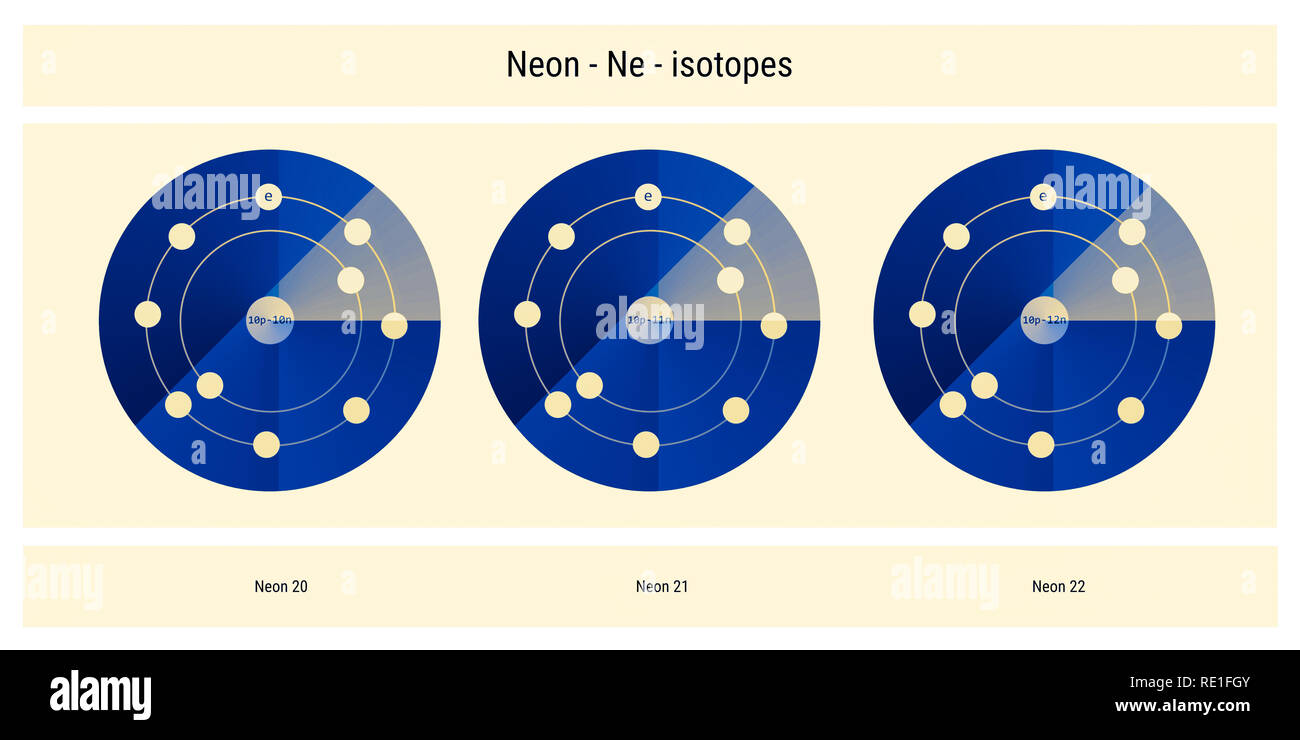
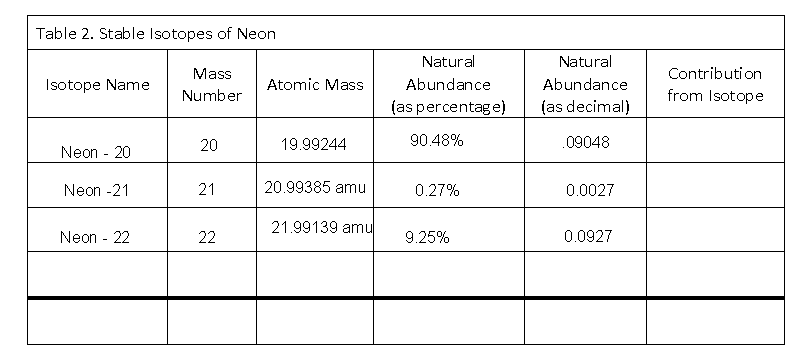
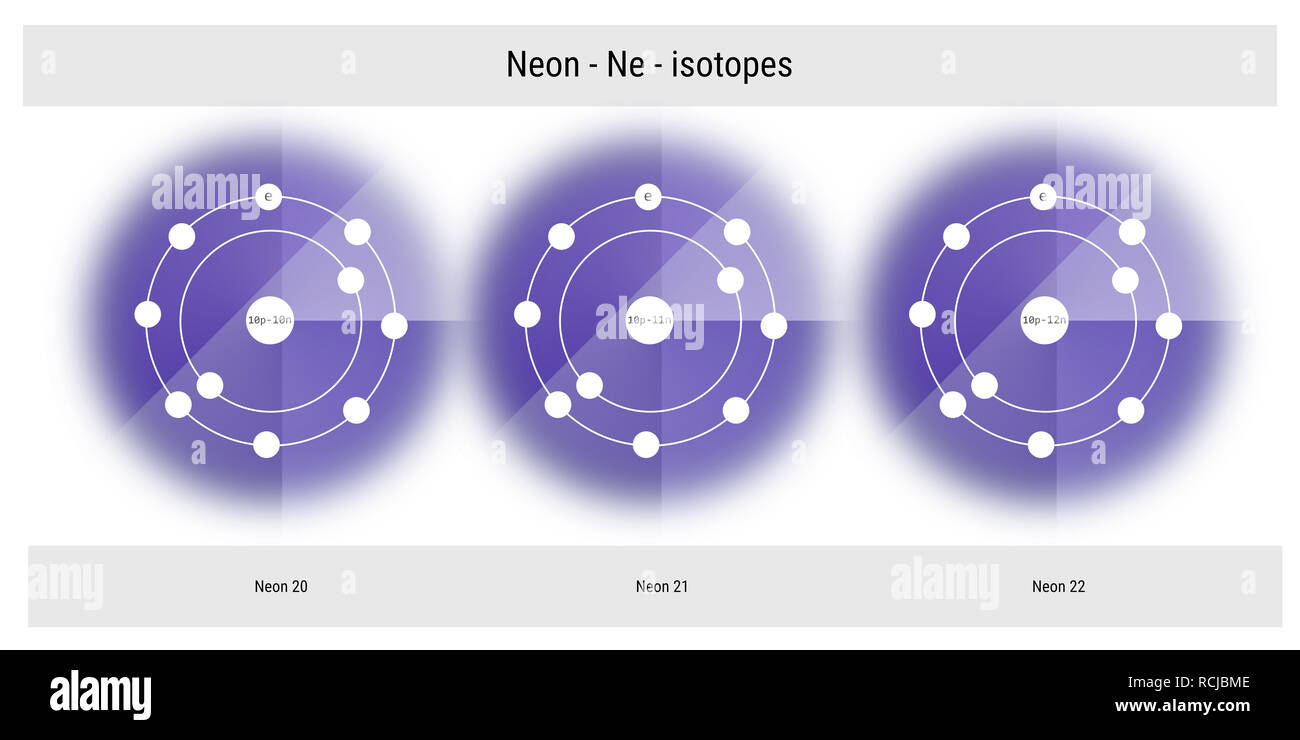
The three stable isotopes of neon Ne^20, Ne^21 and Ne^22 have respective abundances of 90.51% , 0.27 % and 9.22% . The atomic masses of the three isotopes are 19.99 u, 20.99

25 POINTS!!! Write the answers to the following questions. The isotope neon-22 has 1- protons. How - Brainly.com