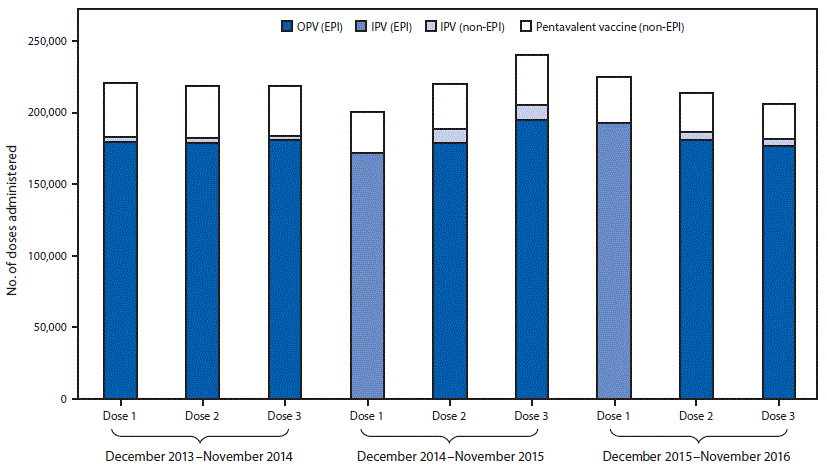
Introduction of Inactivated Poliovirus Vaccine and Impact on Vaccine-Associated Paralytic Poliomyelitis — Beijing, China, 2014–2016 | MMWR
Children in United States vaccinated for polio elsewhere may require revaccination | MDedge Infectious Disease

Evaluating the cost per child vaccinated with full versus fractional-dose inactivated poliovirus vaccine - ScienceDirect

Urea Improves Stability of Inactivated Polio Vaccine Serotype 3 During Lyophilization and Storage in Dried Formulations - Journal of Pharmaceutical Sciences

Fractional dose compared with standard dose inactivated poliovirus vaccine in children: a systematic review and meta-analysis - The Lancet Infectious Diseases