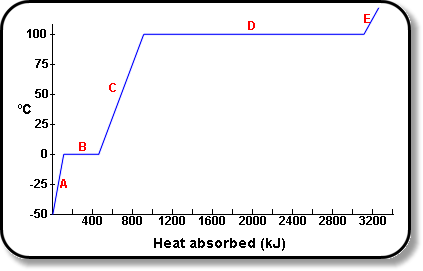
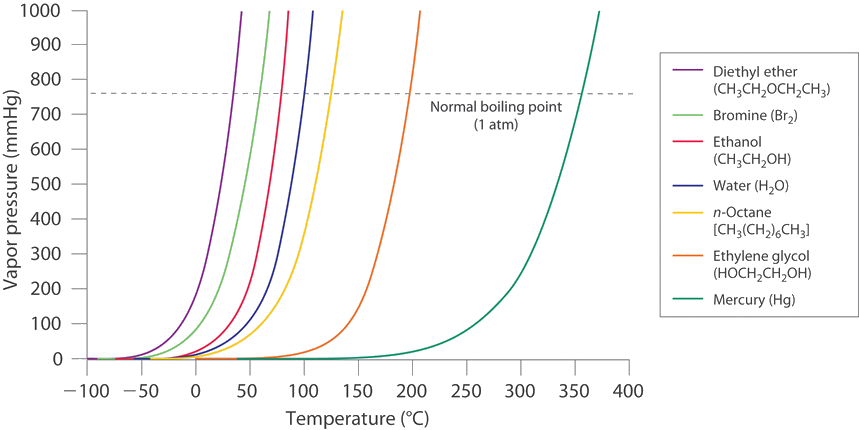
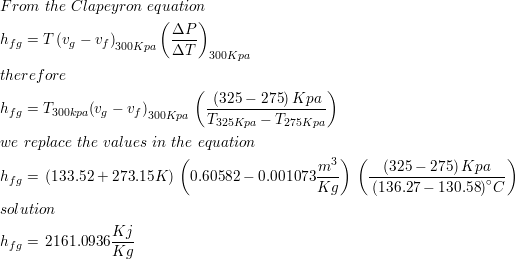SOLVED: The heat of vaporization of. liquid may be obtained from the following equation: p; In p, TT AH = -R R In Pz T -T p (This is an approximated integrated

Ethanol has a heat of vaporization of 38.56 kj/mol and a normal boiling point of 78.4 C - Home Work Help - Learn CBSE Forum

Calculate the enthalpy of vaporisation per mole for ethanol. Given, Δ S = 109.8JK^-1mol^-1 and boiling point of ethanol is 78.5^oC .
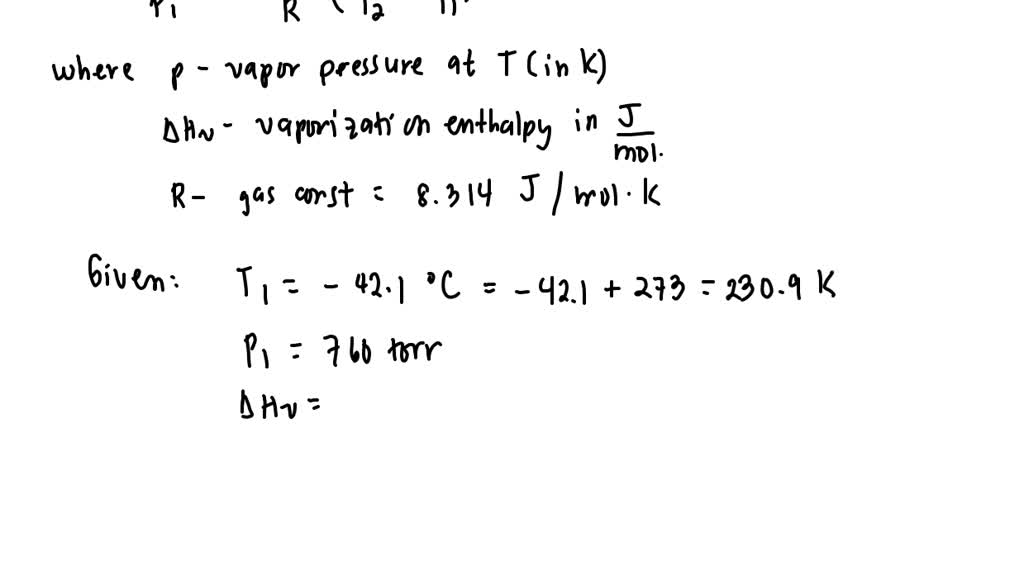
SOLVED: The enthalpy of vaporization of propane is 19.0 kJ/mol and its normal boiling point is -42.1 °C. Using the Clausius-Clapeyron equation, calculate the temperature at which propane has a vapor pressure

Describe (qualitatively) how standard enthalpy and entropy of vaporization of water will change with temperature. | Homework.Study.com

SciELO - Brasil - Enthalpy of mixing and heat of vaporization of ethyl acetate with benzene and toluene at 298.15 k and 308.15 k Enthalpy of mixing and heat of vaporization of


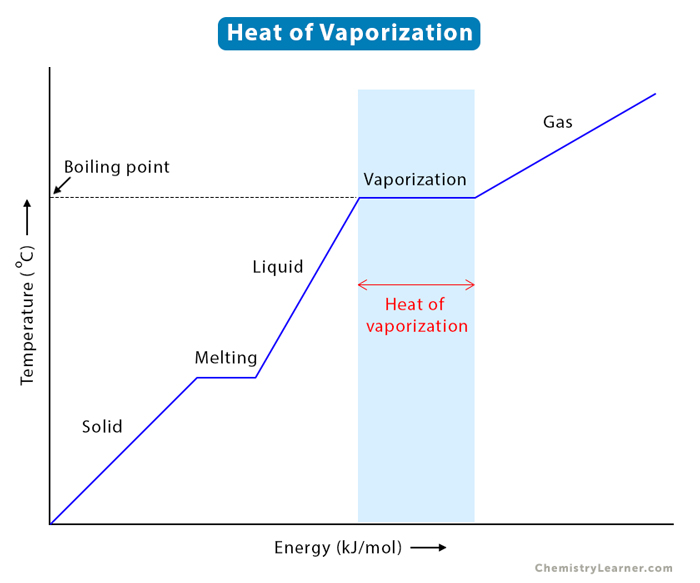

-438.png)