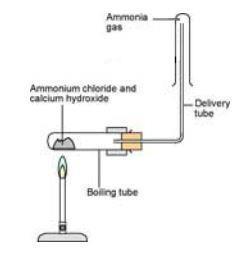
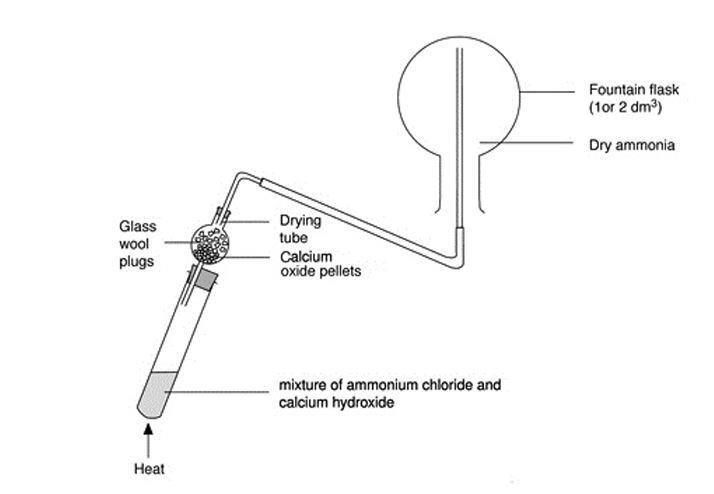
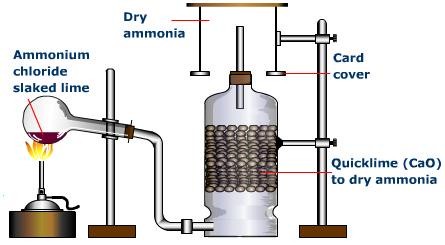
In a gaseous reaction between ammonia and hydrochloric acid (HCl), a white precipitate of ammonium chloride is produced according to the following reaction. NH3(g) + HCl (g) rightarrow NH4Cl(s) Two cotton plugs

Sustainability | Free Full-Text | A New Process for the Recovery of Ammonia from Ammoniated High-Salinity Brine | HTML

G420: Graham's Law of Diffusion – NH3 and HCl Diffusion | Lecture Demonstration Manual General Chemistry | University of Colorado Boulder

If one mole of ammonia and one mole of hydrogen chloride are mixed in a closed container to form ammonium chloride gas , then

Preparation of Ammonia Gas in Laboratory with the Help of Ammonium Chloride and Calcium Oxide Stock Vector - Illustration of white, formula: 220304379

Preparation of ammonia. At left a mixture of ammonium chloride and slaked lime is being heated, this reacts to create ammonia gas. The gas, which is l Stock Photo - Alamy

Phase Equilibriums of Ammonium Chloride Systems as Model Hydrogenolysis Products of Organochlorine Compounds under Naphtha Hydrotreating Conditions | SpringerLink